Until now, SAXS measurements on biological samples were limited to synchrotron facilities or low throughput laboratory setups requiring large sample volumes. The BioXolver with its unique features overcomes these limitations. It offers:
Accelerate your biostructural research
-
High quality data
-
Full automation of multi-sample measurements and data analysis
-
The capability to work reliably with very small volumes of sample
-
Best of all, this capability is available right in your lab, allowing you to shorten your development cycle and accelerate your bio-structural research
Some of our customers



Discovering macromolecular shape and interactions
Using Small angle X-ray scattering (SAXS), the BioXolver collects structural information on biological samples.
The BioXolver can obtain key structural parameters, such as:
- radius of gyration
- molecular weight
- maximum intra-particle distance
- degree of folding
- low-resolution 3D protein shape.
In-lab SEC-SAXS, protein-protein interactions...
Key benefits & features
Publications
Discover how scientists use Xenocs instruments for their research & publications
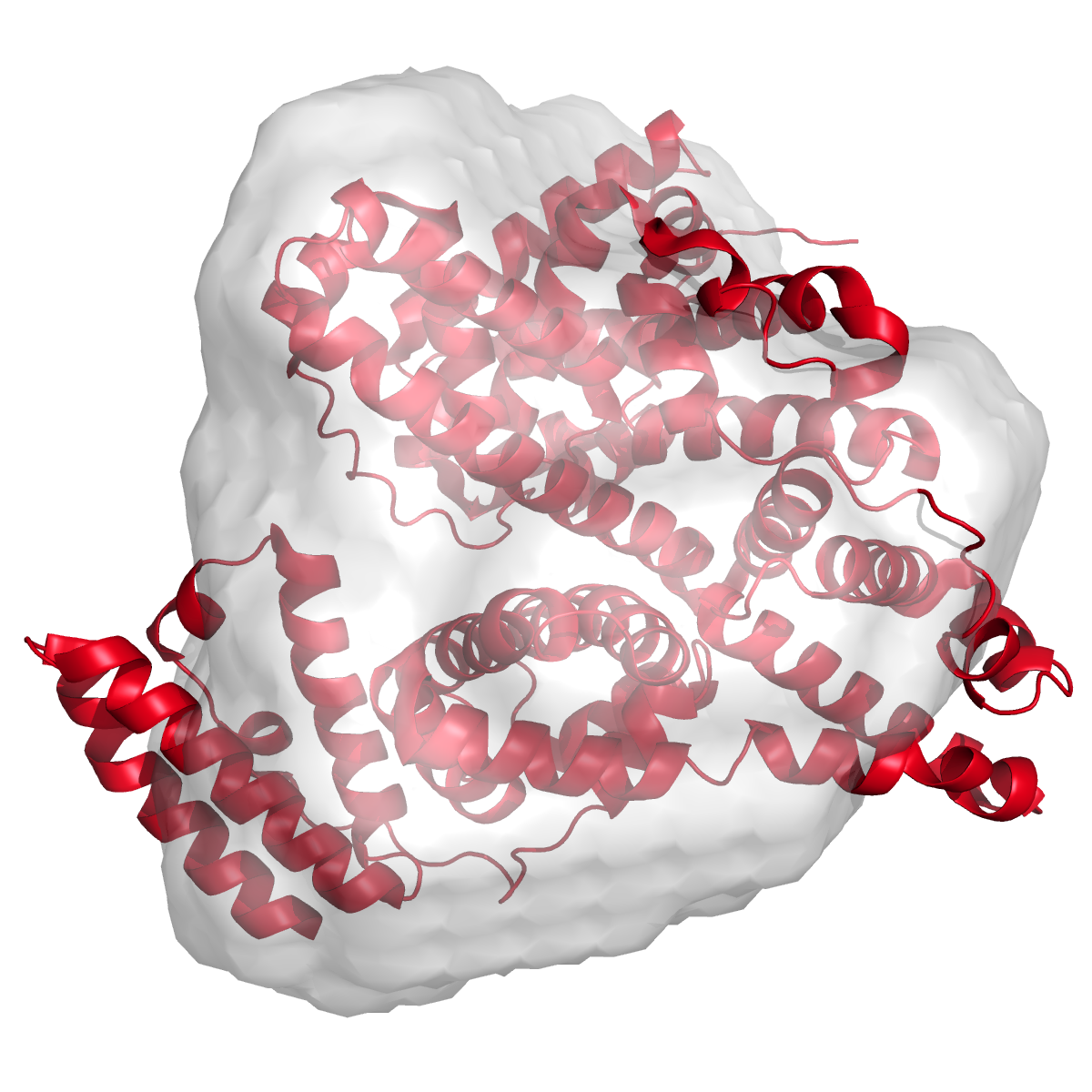
Molecular architecture of the Jumonji C family histone demethylase KDM5B
J. Dorosz, L. Hyltoft Kristensen, N. G. Aduri, O. Mirza, R. Lousen, S. Bucciarelli, V. Mehta, S. Sellés-Baiget, S. M. Øie Solbak, A. Bach, P. Mesa, P. Alcon Hernandez, G. Montoya, T. T. T. N. Nguyen, K. D. Rand, T. Boesen & M. Gajhede
Learn more on benefits & features
High quality SAXS data in the lab
Measurements previously only possible at synchrotron facilities can now be done in the lab.
With the BioXolver you get:
-
Excellent data suitable for modelling and publications.
-
Precision determination of key structural parameters.
-
Structural information of proteins in solutions on a daily basis as complementary information to other characterization techniques.
Due to weak scattering from biological samples, high quality SAXS measurements require an instrument optimized for maximum X-ray intensity and lowest possible background noise. The BioXolver offers a unique combination of both capabilities.
Read more on features
High brightness sources coupled to high quality optics
The BioXolver can be installed with different source options.
It can integrate a MetalJet source to obtain the highest X-ray beam intensity possible in a laboratory instrument.
It can also be installed with the Genix3D, a bright microfocus low maintenance source with intensity levels only previously accessible with high power rotating anode sources. Such performance is achieved by combining the Genix3D long lasting source with new patented FOX 3D multilayer optics having a very high solid angle.
Beamstop-less acquisition with windowless detectors
The BioXolver comes with windowless hybrid photon counting (HPC) detectors which allows for beamstopless measurements.
By operating without beamstop the direct beam is recorded all along the acquisition, which leads to an accurate transmission measurement used to obtain a precise absolute intensity normalization.
Moreover, operating without beamstop reduces the lowest scattering angle achievable as it is no more limited by beamstop size but is sample dependent.
Clean Beam Technology
The BioXolver uses state of the art collimation slits that greatly reduce noise and provide a clean beam.
High quality detection is achieved with hybrid photon counting (HPC) detectors integrated inside vacuum with no interfering window in front of detector sensor (windowless detector). This ensures that weakly scattering samples can also be measured at low concentrations.
Overall measuring background level of the instrument is further improved by proprietary automatic cosmic background removal algorithms suppressing high energy cosmics hitting the detector.
High throughput measurements and results
Full automation from sample to final results on up to 192 samples.
With the BioXolver standard well plates can be loaded & measured automatically using in-line pipetting robot & automated software suite for high user benefits.
-
More samples can be run
-
Short sample-to-sample cycle time
-
Reproducibility of your measurement is insured
Read more on features
Sample loading robot

The advanced pipetting robot avoids cross-contamination of samples through the use of disposable pipette tips, tubeless handling for no sample loss and a fully automated sample cell cleaning and drying.
Automated software package for data analysis

The BioXolver includes powerful data reduction BioXTAS RAW widely used by the BioSAXS community.
It enables standard data operation and data analysis (Rg, molecular weight, etc.), together with advanced analysis using ATSAS plugins.
For high throughput analysis, the automated analysis function analyzes and compiles the results for you.
Low sample volume consumption
Down to 5μL. The lowest sample consumption on the market.
High precision positioning of small sample volumes is ensured by the conjunction of an in-line pipetting robot and the BioCUBE, an advanced capillary flow cell, coupled to machine vision for sample monitoring.
Such low total consumption per sample condition or concentration is highly beneficial for biostructural research.
-
Less precious sample is needed.
-
More experiments can be performed from the same preparation volume.
Read more on features
BioCUBE
Tubeless handling of sample volume down to 5 μl. This is unique on the market.
Each sample is first automatically injected into the BioCUBE measurement cell with the tubeless pipetting robot, then positioned into the beam for measurement with dynamic real-time continuous control of its position.
The sample is maintained at the selected temperature both in the well plate during storage and in the BioCUBE during measurements.
Optionally, the BioCUBE may also be provided with integrated UV/Vis capabilities.
Machine Vision control of 5μl sample volumes

Using machine vision technology, the BioXolver is able to keep a digital eye on the sample at all times.
Once loaded, the sample is positioned with high precision in the X-ray beam while continuous feedback is provided to the automated control software to keep the volume in place.
In-line SEC and UV-Vis combined with SAXS – also in the lab!
Obtain the structure of small and large protein species through in-line gel filtration
Read more on features
Easy switching to SEC-SAXS experiments
Integrated UV-Vis
In situ UV measurement directly in the measurement cell provides concentration information of the sample before and after X-ray exposure or during a SEC-SAXS experiment.
This is especially important in this case as the sample is diluted in the transport path from the (HPLC) column exit to the x-ray exposure position.
































